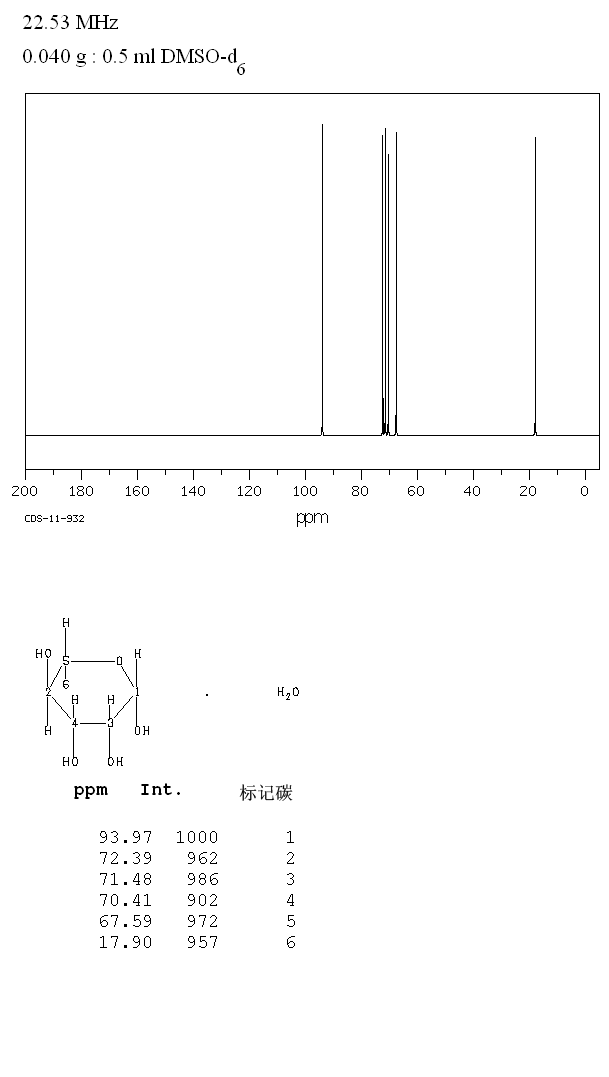
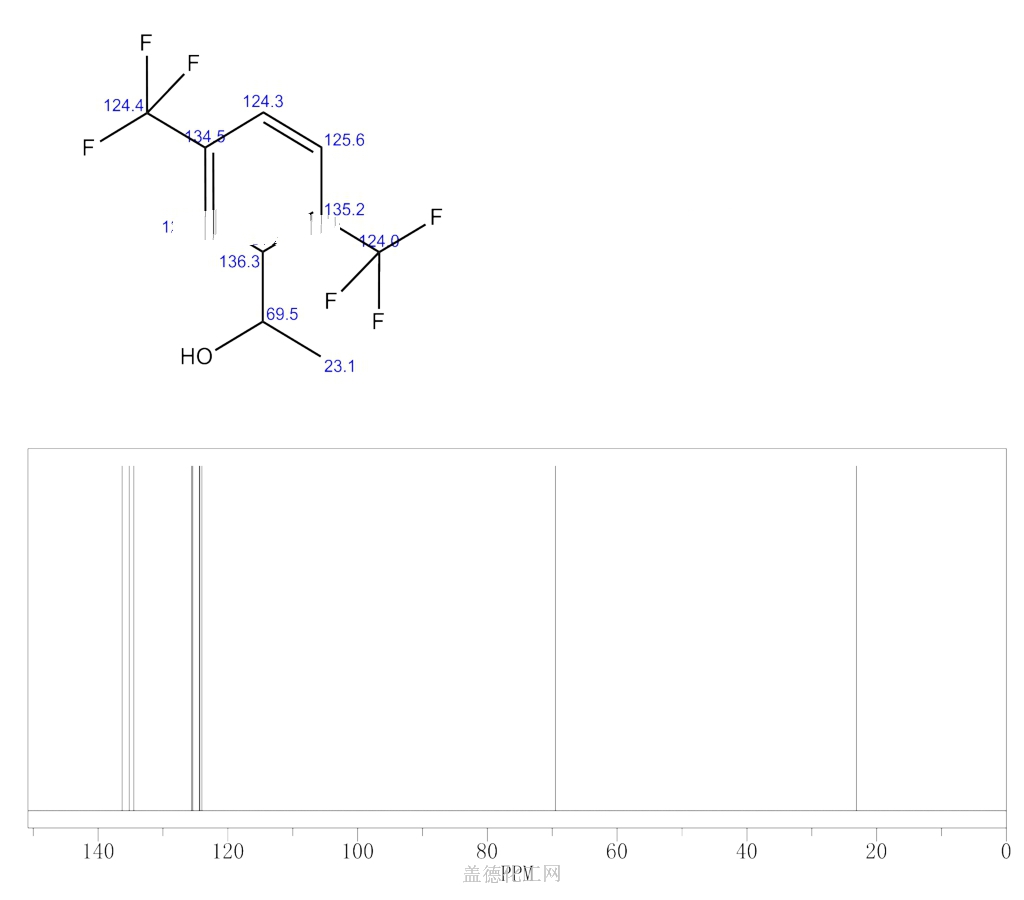
In two-dimensional NMR, the emission is centered around a single frequency, and correlated resonances are observed. Although large amounts of impurities do show on an NMR spectrum, better methods exist for detecting impurities, as NMR is inherently not very sensitive - though at higher frequencies, sensitivity is higher.Ĭorrelation spectroscopy is a development of ordinary NMR. The timescale of NMR is relatively long, and thus it is not suitable for observing fast phenomena, producing only an averaged spectrum. Preferably, the sample should be dissolved in a solvent, because NMR analysis of solids requires a dedicated magic angle spinning machine and may not give equally well-resolved spectra. A disadvantage is that a relatively large amount, 2–50 mg, of a purified substance is required, although it may be recovered through a workup. NMR has largely replaced traditional wet chemistry tests such as color reagents or typical chromatography for identification. Different functional groups are obviously distinguishable, and identical functional groups with differing neighboring substituents still give distinguishable signals. NMR spectra are unique, well-resolved, analytically tractable and often highly predictable for small molecules. The most common types of NMR are proton and carbon-13 NMR spectroscopy, but it is applicable to any kind of sample that contains nuclei possessing spin. Besides identification, NMR spectroscopy provides detailed information about the structure, dynamics, reaction state, and chemical environment of molecules. Similarly, biochemists use NMR to identify proteins and other complex molecules. Detection and analysis of the electromagnetic waves emitted by the nuclei of the sample as a result of this perturbation.The perturbation of this alignment of the nuclear spins by a weak oscillating magnetic field, usually referred to as a radio- frequency (RF) pulse.The alignment (polarization) of the magnetic nuclear spins in an applied, constant magnetic field B 0.The principle of NMR usually involves three sequential steps: As the fields are unique or highly characteristic to individual compounds, in modern organic chemistry practice, NMR spectroscopy is the definitive method to identify monomolecular organic compounds. The intramolecular magnetic field around an atom in a molecule changes the resonance frequency, thus giving access to details of the electronic structure of a molecule and its individual functional groups. The sample is placed in a magnetic field and the NMR signal is produced by excitation of the nuclei sample with radio waves into nuclear magnetic resonance, which is detected with sensitive radio receivers. Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy or magnetic resonance spectroscopy ( MRS), is a spectroscopic technique to observe local magnetic fields around atomic nuclei. Mapping of the locations of these residues onto the helical model observed in the crystal revealed that these two regions are parts of the interior lining of the positively charged helical groove, supporting the hypothesis that the helical oligomer may form in solution.A 900 MHz NMR instrument with a 21.1 T magnet at HWB-NMR, Birmingham, UK In addition, residues in the β-sheet region also showed significant perturbations. Studies of the chemical shift perturbations caused by the binding of single-stranded DNA and mutational analyses have identified the disordered region at the N-termini as the prime site for nucleic acid binding. The NMR structure is almost identical with the previously solved crystal structure, except for a disordered putative RNA-binding domain at the N-terminus. The SAIL protein yielded less crowded and better resolved spectra than uniform 13C and 15N labeling, and enabled the homodimeric solution structure of this protein to be determined. Here, we employed the SAIL method to determine the high-quality solution structure of the SARS-CoV NP CTD by NMR. We have recently developed the stereo-array isotope labeling (SAIL) method to overcome the size problem of NMR structure determination by utilizing a protein exclusively composed of stereo- and regio-specifically isotope-labeled amino acids. So far, the structure determination of the SARS-CoV NP CTD in solution has been impeded by the poor quality of NMR spectra, especially for aromatic resonances. The C-terminal domain (CTD) of the severe acute respiratory syndrome coronavirus (SARS-CoV) nucleocapsid protein (NP) contains a potential RNA-binding region in its N-terminal portion and also serves as a dimerization domain by forming a homodimer with a molecular mass of 28 kDa.
